Why test for Alpha-1?
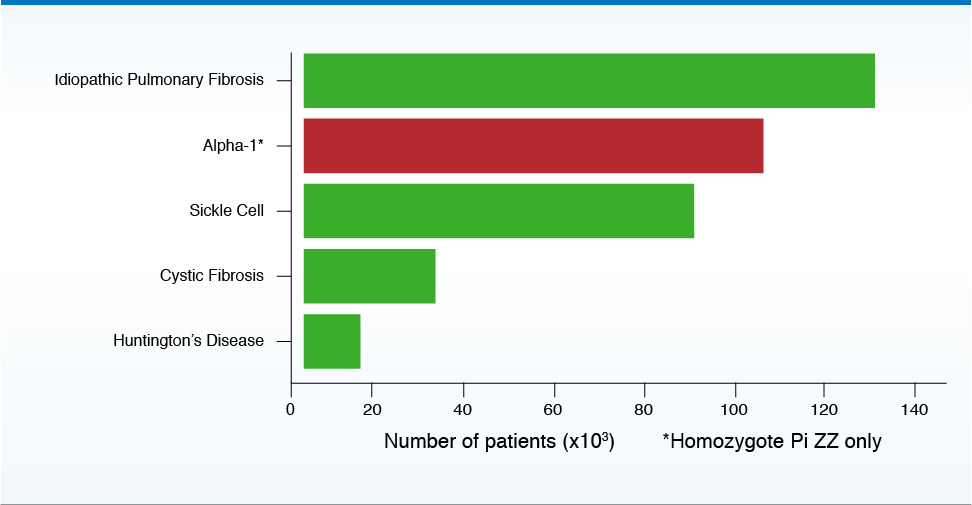
Prevalence of alpha-1 in the United States
-
Alpha-1 is the major known genetic risk factor for chronic obstructive pulmonary disease (COPD), which is the third-leading cause of death in the United States1-3
- Symptoms of COPD include difficulty breathing, cough, mucus production, and wheezing
- Alpha-1 is a potentially fatal genetic form of COPD and may be more common than you think. It can also be found in patients of all ages2,4
- Alpha-1 may be a contributing cause of up to 3% of all COPD cases in the United States5
- Up to 25 million Americans are thought to have at least 1 deficient allele (S or Z), and may pass that gene on to their children6
Alpha1-antitrypsin (AAT) deficiency is a rarely diagnosed disease2
>100,000 people in the United States are estimated to have AAT deficiency5
>90% of those with AAT deficiency (alpha-1) remain undiagnosed6
Leading organizations recommend genetic screening for alpha-1
American Thoracic Society (ATS)7
COPD Foundation8
Global Initiative for Chronic Obstructive Lung Disease (GOLD)9
World Health Organization1
The ATS guidelines recommend screening for alpha-1 in the following7:
- All adults with symptomatic COPD, regardless of smoking history
- All adults with symptomatic emphysema, regardless of smoking history
- All adults with symptomatic asthma whose airflow obstruction is incompletely reversible after bronchodilator therapy
- Asymptomatic patients with persistent obstruction on pulmonary function tests and with identifiable risk factors (eg, smoking, occupational exposure)
- Siblings of individuals with alpha-1
Screening all patients with COPD for alpha-1 can help detect those at increased risk for lung disease
COPD and genetic COPD due to alpha-1 share many of the same symptoms
COPD describes multiple lung diseases, including emphysema and chronic bronchitis. Alpha-1 is often not diagnosed because its symptoms are similar to those of COPD1,7,10:
- Dyspnea
- Decreased exercise tolerance
- Wheezing
- Persistent cough
- Excess sputum production
- Frequent lower respiratory tract infections
- History of suspected asthma
COPD and genetic COPD due to alpha-1 also share many of the same diagnostic tools used to identify them, including11:
- Presentation of symptoms
- Spirometry
- Diffusing capacity
- Chest x-rays
- Chest CT
- Blood gas
An alpha-1 screening kit is the ONLY diagnostic tool that can differentiate COPD from COPD due to alpha-111

IMPORTANT SAFETY INFORMATION
PROLASTIN®-C LIQUID is an alpha1-proteinase inhibitor (human) (alpha1-PI) indicated for chronic augmentation and maintenance therapy in adults with clinical evidence of emphysema due to severe hereditary deficiency of alpha1-PI (alpha1-antitrypsin deficiency).
Limitations of Use
- The effect of augmentation therapy with any alpha1-PI, including PROLASTIN-C LIQUID, on pulmonary exacerbations and on the progression of emphysema in alpha1-PI deficiency has not been conclusively demonstrated in randomized, controlled clinical trials
- Clinical data demonstrating the long-term effects of chronic augmentation or maintenance therapy with PROLASTIN-C LIQUID are not available
- PROLASTIN-C LIQUID is not indicated as therapy for lung disease in patients in whom severe alpha1-PI deficiency has not been established
PROLASTIN-C LIQUID is contraindicated in immunoglobulin A (IgA)-deficient patients with antibodies against IgA or patients with a history of anaphylaxis or other severe systemic reaction to alpha1-PI products.
Hypersensitivity reactions, including anaphylaxis, may occur. Monitor vital signs and observe the patient carefully throughout the infusion. If hypersensitivity symptoms occur, promptly stop PROLASTIN-C LIQUID infusion and begin appropriate therapy.
Because PROLASTIN-C LIQUID is made from human plasma, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. This also applies to unknown or emerging viruses and other pathogens.
The most common adverse reactions during PROLASTIN-C LIQUID clinical trials in >5% of subjects were diarrhea and fatigue, each of which occurred in 2 subjects (6%).
Please see full Prescribing Information for PROLASTIN-C LIQUID.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
References:
1. World Health Organization. α1-antitrypsin deficiency: memorandum from a WHO meeting. Bull World Health Organ. 1997;75(5):397-415. 2. de Serres FJ. Alpha-1 antitrypsin deficiency is not a rare disease but a disease that is rarely diagnosed. Environ Health Perspect. 2003;111(16):1851-1854. 3. American Lung Association. How serious is COPD. http://www.lung.org/lung-health-and-diseases/lung-disease-lookup/copd/learn-about-copd/how-serious-is-copd.html. Accessed October 1, 2018. 4. National Institutes of Health. Alpha-1 antitrypsin deficiency. Genetics home reference. http://ghr.nlm.nih.gov/condition/alpha-1-antitrypsin-deficiency. Accessed April 17, 2014. 5. Campos MA, Wanner A, Zhang G, Sandhaus RA. Trends in the diagnosis of symptomatic patients with α1-antitrypsin deficiency between 1968 and 2003. Chest. 2005;128(3):1179-1186. 6. de Serres FJ, Blanco I, Fernández-Bustillo E. Genetic epidemiology of alpha-1 antitrypsin deficiency in North America and Australia/New Zealand: Australia, Canada, New Zealand and the United States of America. Clin Genet. 2003;64(5):382-397. 7. American Thoracic Society/European Respiratory Society. American Thoracic Society/European Respiratory Society statement: standards for the diagnosis and management of individuals with alpha-1 antitrypsin deficiency. Am J Respir Crit Care Med. 2003;168(7):818-900. 8. COPD Foundation. The diagnosis and management of alpha-1 antitrypsin deficiency in the adult. https://journal.copdfoundation.org/jcopdf/id/1115/The-Diagnosis-and-Management-of-Alpha-1-Antitrypsin-Deficiency-in-the-Adult. Accessed October 1, 2018. 9. Global Initiative for Chronic Obstructive Lung Disease. Pocket Guide to COPD Diagnosis, Management, and Prevention. 2010:1-28. 10. Learning about alpha-1-antitrypsin deficiency (AATD). National Human Genome Research Institute. https://www.genome.gov/19518992/. Accessed August 15, 2014. 11. Data on file, Alpha-1 Genetics Laboratory.


